The entire universe is made up of small particle like elemental structure which are combined together by some chemical bonding called . These are called as molecules. The molecules are formed when two or more atoms are combined together. Every substance in universe is made up of molecules only.
Molecules:
The molecules give the substance a particular size, shape and have its own characteristics. Different kinds of atomic elements are combined with the molecular structure to form a new molecular structure. For example 2 hydrogen atom and an oxygen atom is combined with chemical bonding to form H2O ( water).
Atoms:
The word atom is derived from Greek word “ATOMLO” which means indivisible. The term autumn was proposed by John Dalton. Matter is made up of molecules and molecules are made up of atoms. Atoms are fundamental building blocks of matter.
Atomic Structure:
In the atomic structure there are three basic subatomic particles; electron, proton and neutron which form the building blocks of all atoms and those are called fundamental particles.
Electron:
It is a subatomic particle which carries a unit of negative energy. It is discovered by Sir JJ Thomson during the study of cathode rays in the discharge tube. The name was introduced by Stoney.
e/m of electron = 1.76*108 c/g
Proton:
It is a subatomic particle which carries a unit of positive energy. The name Proton was suggested by RutherFord.
e/m of Proton = 9.58*10⁴ c/g
Neutron:
It is a subatomic particle which carries no charge. It is discovered by Chadwick.
Mass of neutron= 1.675*10-²⁴g
Protons and neutrons are found in the nucleus at the center of the atom. The electrons revolve outside the nucleus in the shells for energy levels.
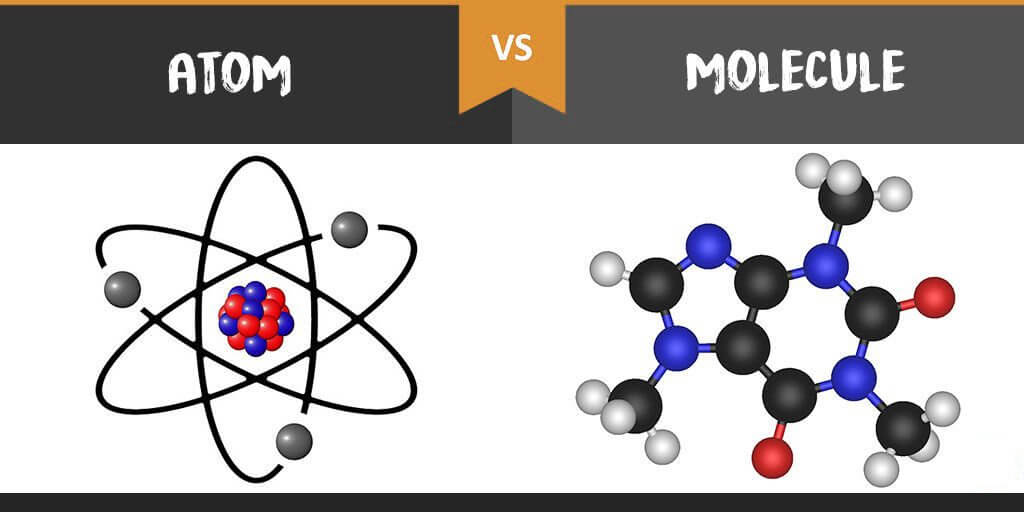

Difference between Atoms and Molecules:
| Atoms | Molecules |
|---|---|
| Size | |
| Atoms are the smallest substances in any element. | Molecules are also smaller substance in the elements but when compared to atoms it is big |
| Components | |
| Atoms are made up of protons, electrons and neutrons | Molecules are made up of two or more atoms combined together with chemical bonding |
| State Of Existence | |
| Atoms may not exist freely. | Molecules exist in a free state. |
| Shape | |
| Atoms generally have a spherical shape. | Several atoms are combined together to form a molecule which gives the molecule angular or linear or triangular shape. |
| Bond | |
| It is a Nuclear Bond | It is a covalent bond. |
| Visibility | |
| Atoms are generally not visible. | Molecules are also not visible but can be seen through a magnifying microscope. |
Conclusion:
When all the atoms and molecules are combined together and work properly then only an element can be formed perfectly. When two or more atoms from a different element combined together with chemical bonding a new molecule is formed with new properties.
For further reading about Difference between Centripetal Force and Centrifugal Force click here.


